Happy Monday, everyone! Don’t be fooled – I myself am not overly enthused about it being Monday. But we can pretend we are nonetheless! Of course, since it is Monday, I have come bearing a new blog for you. This one is about batteries – and it is somewhat technical and long, though my goal is to try and cater to an audience who may not have any technical expertise whatsoever. Another goal of mine is to try and make this post somewhat fun and enjoyable. It is important for me to share my experience in this topic because batteries play such a huge role in our lives, but I have found that most people don’t have a great understanding of them. There is no denying that battery equipment – especially vehicles – are becoming more and more prevalent in commercial and industrial applications. After all, my job only exists because of the emergence of battery vehicles in the mining industry. So if you happen to be someone who has never had the opportunity to learn about batteries, then I sincerely hope this post is enlightening for whatever context in which you may be using them.
Firstly, why do I have any authority to be educating anyone about batteries? Well, I have spent 4 years working with & learning about batteries. I try to keep myself knowledgeable of contemporary battery research by reading a new academic study every week, and I’ve done some minimal research myself. At this point I’m convinced that batteries are the only thing I truly know anything about. I also have a battery-inspired tattoo, so if that’s not sufficient enough then I’m not sure what would be! That, of course, was a joke – but the tattoo is still very real. All of the batteries I’ve worked with so far have been lithium-ion, which is what I’ll be focusing on today. However, a substantial portion of this post can be applied to all kinds of battery “flavours”.
The primary purpose of a battery is to store electrical energy for a (relatively) indefinite amount of time, until needed. Batteries have other purposes too; For instance, when a battery is dead it may be useful as a bookend. All electrical energy is sourced from other types of “potential” energies. For instance, a hydroelectric dam uses gravitational potential energy (the energy associated with water falling because of gravity) and transforms it into kinetic energy (falling water rotates a turbine) which is then transformed into electrical energy via a generator, which entails some complex electromagnetic mechanisms that aren’t necessary to explain here. Batteries, and any other energy providers are no different. Batteries use the chemical potential energy stored in its reactants and transforms it into electrical energy, which only happens when an electric field is present within the battery (ie. a “voltage difference“). Something to remember when reading the rest of this post: “electricity” can be aptly summarized by the pursuit of electrons trying to get to the most comfortable place they can find, by taking the easiest route as possible. The most comfortable place an electron can go is to a place of higher voltage, and it does so by means of the path of least resistance. Since electrons posses electrical charge, they interact with electric fields which “cause” them to move through these paths; and to put it simply, movement = energy. That is how we exploit them for power!
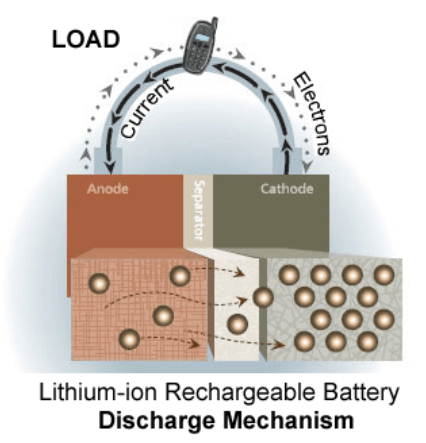
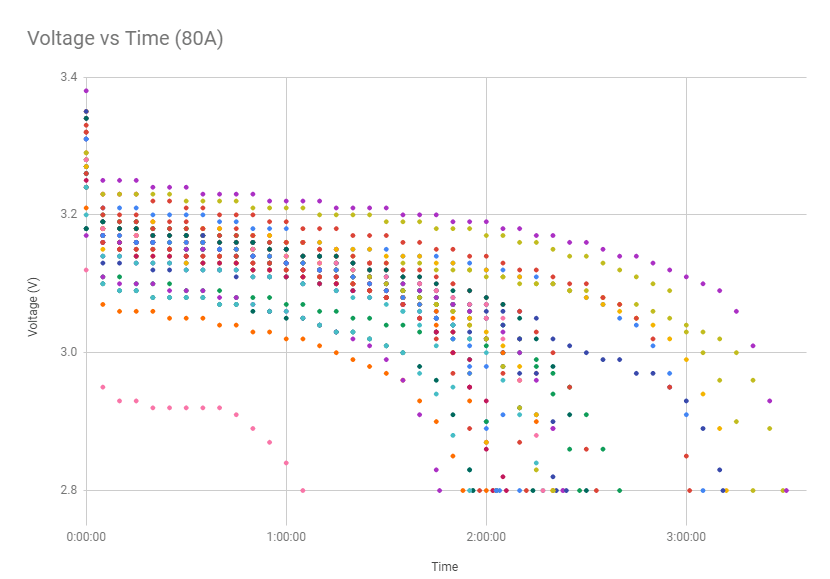
Battery cells (the fundamental constituents of a battery) are composed of 3 distinct components: The electrodes (anode & cathode) and electrolyte. The primary mechanism which drives battery operation is the electrodes having a different “charge” from one another (one has an abundance of free electrons, the other doesn’t), meaning there is a “potential” between them (this is what electricians would call a “voltage” across the electrodes). The amount of “voltage” depends on how many more free electrons that one electrode has compared to the other. When a load (say, a lightbulb) is connected to a battery, it must act as an external path from one electrode to another. The electrodes are separated by a separator within the cell so that they can’t internally exchange electrons; they must do so externally. When this external path is complete, electrons will leave the negative electrode (electrons that are all bunched up in the negative electrode will want to “break free” as soon as they have the chance, since they are all repelling from each other). This external path carries the electrons through the load (lightbulb) and to the positive electrode (higher voltage), energizing the lightbulb and it will light up! As the cell discharges and the electrons move from the negative electrode to the positive, the potential difference between the two lessens as well (since there is less of an abundance of electrons creating that potential in the negative electrode), which means the cell’s voltage will reduce. The graph below is data I collected during the summer of 2019 which demonstrates the cell voltage fade throughout a discharge cycle. This is how we know the difference between a fully charged battery and an almost dead one: we measure the voltage between the electrodes. Note: the most annoying thing about all of this is that electrical current is conventionally viewed as travelling the opposite direction to electrons, flowing from positive to negative. In reality, nothing is travelling in that direction during this part of the process.
What does the electrolyte do? While this electron-roadtrip takes place, the electrolyte carries positively charged particles from the anode (negatively charged electrode) to the cathode. The electrolyte is paired with a separator that prevents the electrons from internally travelling to the other electrode, but it does not inhibit the positive particles. Once all of the positively-charged particles pair up with electrons in the cathode, we say the battery is at a 0% state-of charge (SOC). In order to re-charge the battery, an external power supply must be connected to the battery with an “over-voltage” such that the electrons want to travel through the power supply back to the anode, in order to repeat the process all over again.
Of course, this is only half the story – we have not really discussed the “chemistry” part of electrochemistry. I won’t delve too much into that aspect since the process isn’t so universal from battery to battery; although, it is important to remember that all of the mechanisms discussed here are driven by chemical reactions that happen within the battery. For lithium-ion batteries, these “positive particles” I keep referring to are lithium ions. In a lead-acid battery, it’s hydrogen ions. Another thing to note about li-ion batteries in particular is that there are many different kinds of li-ion batteries. The chemistry of the anode and cathodes are what give different “flavours” of lithium-ion batteries. For instance, I primarily work with lithium iron phosphate batteries (LFP). The cathode is made up of lithium iron phosphate (LiFePO4), and the anode is made up of graphitic carbon. The electrolyte is made up of ethylene carbonate–dimethyl carbonate and lithium perchlorate. Or in other words, as most physicists would say: it’s all chemicals, who cares!

Why are there different “flavours” of li-ion batteries? Each “flavour” has its strengths and weaknesses. The cells that the vehicles in our mine use (LFP) are much more thermally resistant than other li-ion chemistries – which is super important in a hot mine like the one I work at. LFP also has high specific power (max power output per unit mass), which is useful in the context of 40-ton trucks who require lots of power to haul rock up a ramp. The tradeoff is that LFP cells have low specific energy (energy capacity per unit mass), meaning that we need a lot more LFP cells than we would, say, NMC cells (Lithium Nickel Manganese Cobalt Oxide) to ensure that our vehicle will last a sufficient amount of time. A lot of other mines use NMC cells in their battery equipment (mostly service vehicles) but it tends to be a challenge to keep them thermally stable in hot & humid environments. A thermally unstable li-ion battery can lead to gaseous venting or even a fire. Tesla uses NMC chemistry in their vehicles. Apple iPhones use a hybrid Lithium Cobalt Oxide (LCO) and Lithium Polymer (LiPo) chemistry for their batteries. You can read all about the different types of li-ion chemistries here.
How do we measure the characteristics of a battery? Since I always work with the same type of cell, the most important characteristic of a cell I deal with is its capacity. I measure this in ampère-hours (Ah), although capacity is generally measured in watt-hours (Wh), which is a more genuine energy capacity unit. Watt-hour units consider both the voltage and the current associated with the battery, although amp-hours are also a decent measure since a battery’s voltage stays fairly constant as a device consumes its power. The batteries I work with are around 280Ah brand new, whereas the standard Alkaline Energizer E91 AA battery is about 2-3Ah. The most accurate way to measure a li-ion cell’s capacity is by brute force – you must charge it completely, then discharge it completely. Doing this allows you to measure the amount of time it takes to deplete the battery at a constant current, and that time multiplied by the current value will give you the cell’s amp-hour rating. For instance, if a li-ion cell lasts 2 hours connected to a constant 50A load before dying then we know it’s capacity is 2h*50A = 100Ah.
Well, there you have it. There aew so many more things to know about the fundamentals of batteries – especially the lithium ion variety – but I think I’ve addressed quite enough of it here. Batteries will be a substantial part of the world’s energy infrastructure in the future, though they have already made quite an impact in the automotive industry. I have lots of thoughts on the future of battery vehicles in the mining industry as well as the technical side of things, but I’ll have to save that for another blog. Thank you for making it to the end of this post, I hope you’ve learned something along the way!
(October 19th, 2020)
Further Reading:
1https://batteryuniversity.com/
2https://www.science.org.au/curious/technology-future/batteries