It’s been a while since I last wrote on here – life at Sandvik has been quite busy. For those who are new to astronomine, here’s an introduction: I’m Haley Blinn, a Battery Electric Vehicles (BEV) Applications Engineer at Sandvik. I am 25 years old, and battery electric mining vehicles have been the backbone of my entire (albeit short) professional career so far. I started this blog in June 2020 to process and discuss my thoughts as I navigated my way within the mining industry. Throughout my short career, I’ve found two new passions – battery technology and mining; although, what hasn’t changed since joining the industry is that I am most passionate about learning. I use this platform to do what I do best – ask questions about complex topics, and hopefully learn some new things along the way. Simultaneously, I use this platform to attempt what I do worst – articulate what I have learned to you. Let’s get to it!
Working for a manufacturer of BEVs has opened my eyes to the countless challenges that a new technology brings to any industry – let alone mining, where the application is inherently a complex endeavor. In many ways, introducing BEVs has caused a re-write of the conventional understanding we have of mining. What implications do BEVs have on safety? Mining methods? Productivity? Mine Infrastructure? Maintenance? Operating costs? As the manufacturer, where does our responsibility lie in the education of all of this? Of course, all these topics can be written about at length, which I intend to do so at some point – but I figured the most natural place to begin is at the end. That’s where I started in this industry, after all. What happens to a battery at the end of its life?

“The end” is chronologically the last component of a battery’s story; although for me, it’s where the conceptual argument for BEV technology begins. A primary argument in favour of transitioning to BEV technology is that it is more eco-friendly than fuel-based alternatives. This is true in the sense that BEVs themselves are not spitting out harmful emissions into the atmosphere wherever they are driving. In addition to this, we often toss in “sustainability” as another benefit of BEVs; although, if we operate BEVs under the same principle that we do conventional fuel-based vehicles (ie. treating batteries as an expendable energy source to be eventually disposed of) we’re not completing the “circle” part of circularity – this, by definition, is not sustainable. Sure, perhaps we’ve eliminated tens of thousands of liters of diesel over the life of a mining BEV (which is a step in the right direction!); However, mining as an industry is not sustainable – at some point it will not be possible to take any more stuff from the inside of this planet as there won’t be anything of value left. This includes the stuff we use to make batteries – That is, until we find an economical way to mine elsewhere in the universe. The mining industry won’t become obsolete in my lifetime, and probably not for a very, very long time – but I think discussing this topic is valuable. Reducing emissions is a great start towards achieving global environmental stability; but the prolonged existence of our global ecosystem requires energy sources as renewable & reusable as we can possibly get. As such, in an ideal world, there is no end to a battery’s useful life – just a new beginning.
How far away is a global lithium crisis?
Generally, when discussing BEV batteries today, we are referring to lithium-ion batteries – the most contemporary industrialized rechargeable battery technology. So, understanding the economics of BEVs necessitates discussion of lithium or other li-ion supporting minerals. According to an analysis done by EY in April 2022, global lithium production must quadruple from 2021 to 2030 to 2 million tons produced per year to meet growing demand otherwise there will be a significant market deficit. In 2021, global lithium production was 490 thousand tons. Similar trends indicating a growth in future demand can be found amongst many of the minerals required to support global electrification, such as copper and nickel. According to the 2022 U.S. Geological Survey (USGS) mineral commodity summary on Lithium, worldwide lithium resources are totaled at 89 million tons. However, it’s important to note that not all these resources are economically recoverable or feasible to extract; and conversely, these only reflect known resources which is subject to grow with more discoveries. Though, if we continue under these assumptions, a simple calculation shows that lithium resources could be exhausted within 45 years. I will (hopefully) still be in the workforce at that time, though perhaps writing blog posts about nuclear EVs instead. Given this, as the demand for lithium-ion batteries grows and concerns about the environmental impacts of mining and battery disposal increase, lithium-ion recycling will become more critical.
According to a report published by the International Energy Agency (IEA) in 2020, only about 5% of the lithium-ion batteries reaching end-of-life were recycled in 2019, and the recycling rate is expected to increase to only 18% by 2030 without significant policy intervention. So, in summary, we have plenty of lithium reserves left for the medium-term future which is one of the reasons we are not very good at recycling li-ion batteries today.
How is battery-grade lithium produced from natural resources?
The contents in the following section are a summary of C. Tabelin et al. (2022) – a great resource for learning the status of lithium resources and associated mining processes. To begin answering the question, it is important to know that there are different types of lithium-ion batteries, and further, different natural occurrences of lithium. Lithium is found primarily in 3 different contexts: salt lake brines, spodumene ore within the earth’s crust, and in hectorite – a soft clay material.
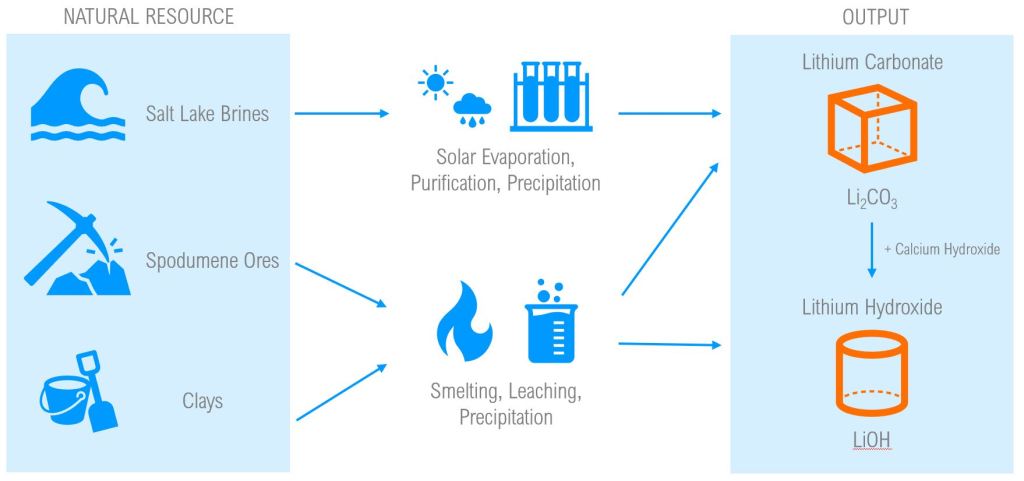

Most of the world’s identified lithium can be found in salt like Li-bearing brines – However, these brines contain low lithium concentrations relative to most naturally occurring hard-rock deposits (ie. spodumene ore deposits). In comparison to hard-rock mining, brine processing facilities generally require lower exploration costs, lower capital costs, and lower operating costs. Although, these brines possess very low lithium compared to other coexisting ions like sodium, potassium, calcium, and magnesium, which leads to challenges in lithium yield efficiencies when processing brine using traditional purification techniques like solvent extraction and precipitation. Better techniques to increase lithium yield in brine processing are still being developed and not yet industrialized.
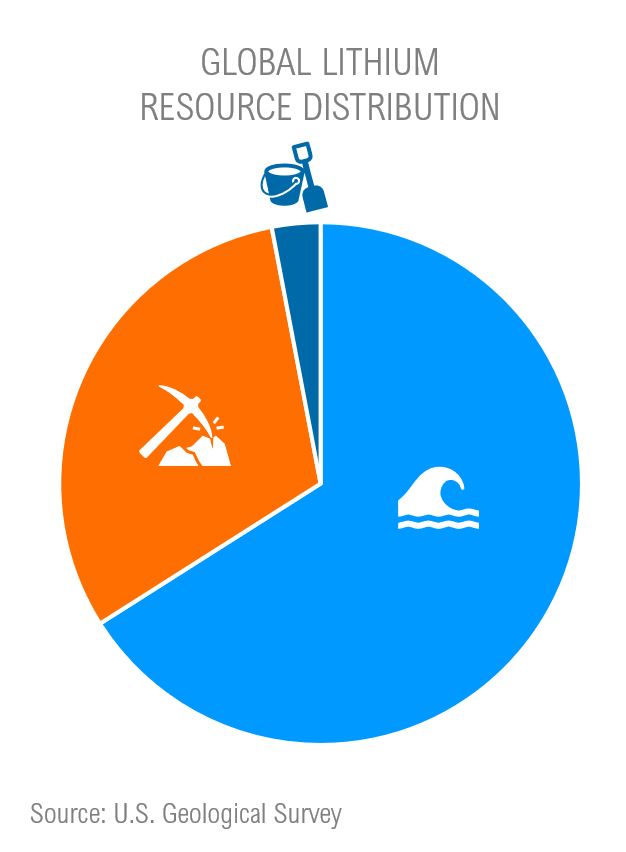
Hard rock and hectorite clay mining methods are of economic interest since they span a wider geographic footprint compared to li-bearing salt lakes, and as mentioned above, generally consist of more concentrated lithium content than li-bearing salt lakes. Of course, hard rock and clay mining are more energy intensive and expensive endeavours; in particular, underground hard-rock mining imposes high exploration and infrastructure costs – but I can’t say I know anything about that 😉.
The primary products of lithium processing for battery manufacturing are Lithium Carbonate (Li2CO3) and Lithium Hydroxide (LiOH). While these two substances have varying chemical properties and economic value, both are used in the fabrication of Li-Ion batteries, alongside other minerals.
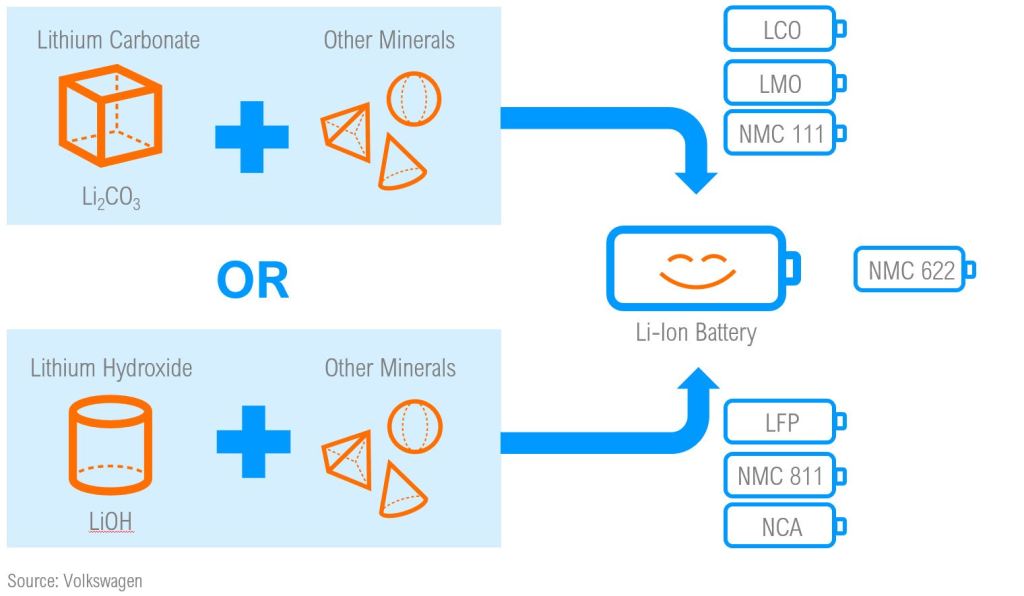
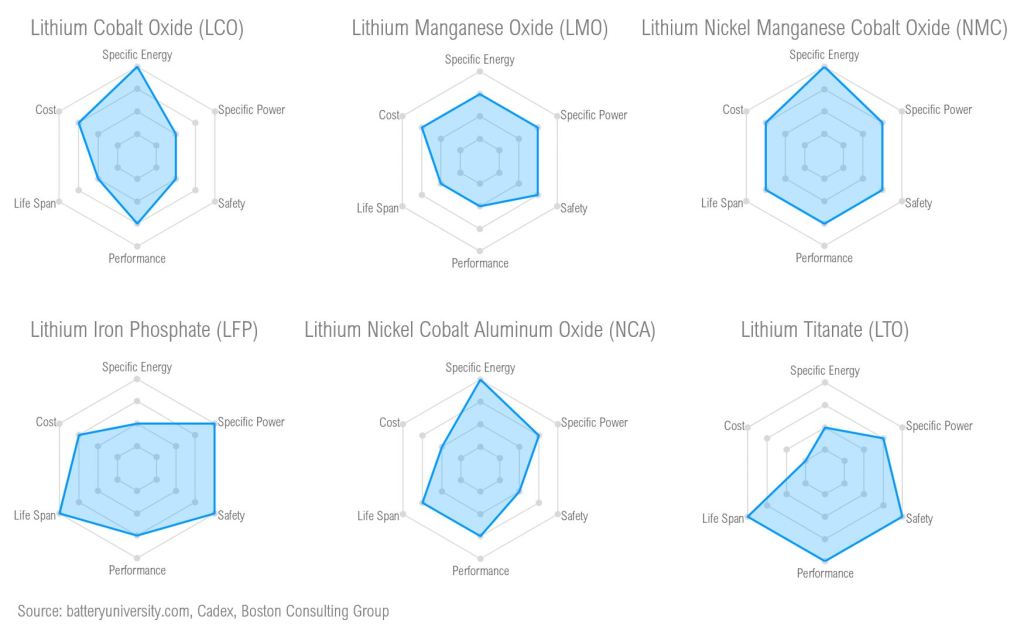
Not all li-ion batteries are the same, there are several li-ion subchemistries that are used in different applications. For instance, Lithium Cobalt Oxide (LCO) is most often used in personal electronic devices, whereas Lithium Nickel Manganese Cobalt Oxide (NMC) is the basis of the new Tesla 4680 battery. Generally, the differences in li-ion subchemistries primarily manifest within the cathode chemistry – an internal component to the li-ion cell. Some li-ion subchemistries require the use of Lithium Hydroxide, while others use Lithium Carbonate.
How much emissions are associated with the fabrication of a li-ion battery?
The amount of energy consumed and emissions involved in the development of a Li-ion cell depends on the type of resource (salt lake, spodumene ore, hectorite clay) the lithium was extracted from, and what type of cell is being made. Life cycle analyses performed by J. Kelly et al. (2021) discuss the difference in energy use, GHG emissions, and water consumption between the production of NMC811 and NMC622 cells. The study considers several facets of the process including lithium acquisition; lithium concentration; production of lithium chemicals, battery cathode powders, and batteries; and associated transportation activities along the supply chain. In this study, it was found that battery-grade lithium compounds produced from salt lake brines had less life cycle GHG emissions and freshwater consumption per tonne than if mined from ore-based resources. Also, cells produced from LiOH yield marginally lower GHG emissions than those fabricated from Li2CO3.
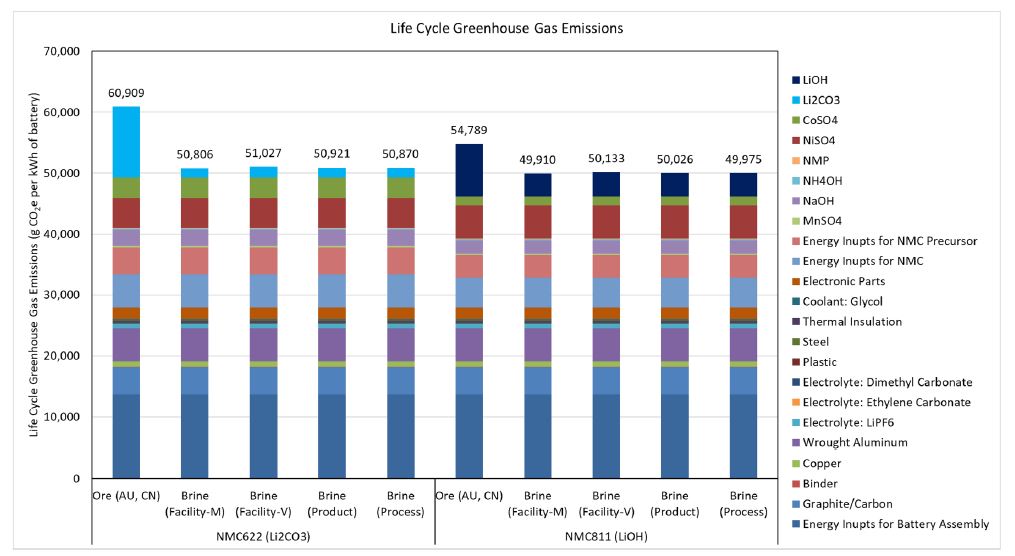
The chart above shows that the fabrication of a Li-ion battery yields ~50kg CO2 produced per kWh of battery, which includes mineral transportation and external-to-cell battery components. These results will vary from battery pack to battery pack; for instance, a Sandvik truck battery pack is comprised of different ratios of telemetry devices, packaging, thermal insulation, and more, as compared to a Tesla Model Y battery pack per kWh of battery. Generally, a commercial passenger BEV’s battery ranges from 20 kWh – 100 kWh, meaning that the production of the battery would be on the order of 1,000kg – 5,000kg CO2 produced when fabricating a battery pack.
For reference, burning 1 litre of gasoline produces ~2.3kg of CO2, and burning 1 litre of Diesel fuel produces 2.7kg CO2. For the standard car, burning a single 45L tank of gasoline produces 103.5kg CO2, which means that 10-50 full tanks of gas burned will match the kg CO2 produced in developing a BEV battery. It is also important to note that this calculation does not consider the GHG emissions associated with the extremely energy-intensive process of extracting and refining oil to produce gasoline; on the other hand, this also does not consider the GHG emissions associated with the energy required to recharge a BEV battery – although that figure will vary by energy source. In the US, the average electricity production emissions is 0.371kg CO2 per kWh. According to this analysis by Innovation Origins, one litre of gasoline from well to tank produces 0.720kg CO2 (before being consumed at the vehicle), although this varies depending on the region the oil is extracted and refined.
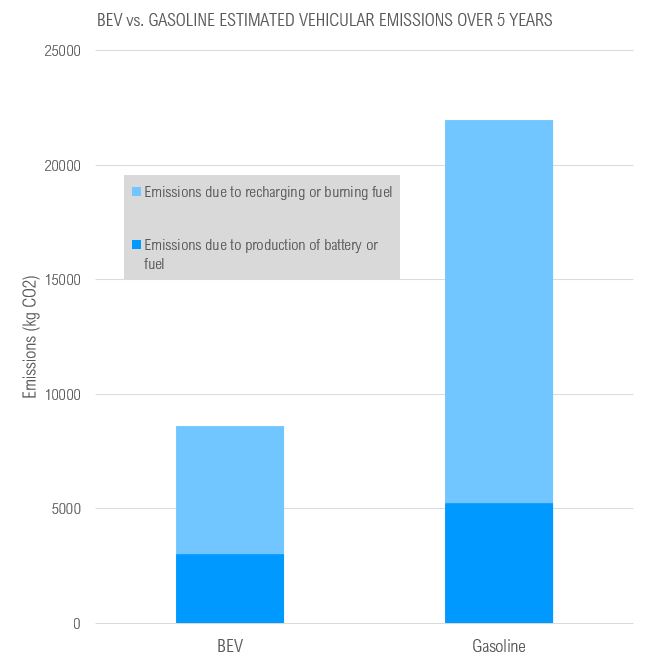
Given this information, the chart above illustrates the estimated GHG emissions associated with the use of passenger vehicles over a 5 year period. Assumptions are drawn from Natural Resources Canada 2023 Fuel Consumption Guide, including averages of 11km/L for gasoline vehicles, 5.3km per kWh for BEVs, and 16,000km driven per year. In conclusion, 80,000 km travelled (16,000km/year) plus fabrication emissions results in ~2.5x CO2 emissions for a gasoline commercial vehicle as compared to a BEV.
It is important to note that the study by J. Kelly et al. (2021) does not consider the GHG emissions associated with any end-of-life energy analysis for battery packs and cells (ie. disassembling, disposing, recycling, etc). The reason for this becomes quite apparent if you search for figures related to battery recycling – there simply is not a standard, unified, and industrialized recycling process when it comes to Li-ion cells.
How are Li-Ion batteries recycled?
The contents in the following section are largely based on results from J. Mao et al. (2022) – a highly recommended read if you are interested in present-day Li-ion recycling methods and challenges. The current status of Li-ion battery recycling is not obvious to the average user of Li-ion battery products – it is an extremely dynamic industry whose sub-processes are in varying stages of industrialization. Many people are aware that battery recycling exists, so that knowledge may lead to the assumption that there is an end-to-end recycling solution for the batteries in all EVs – but this is not the case. My primary takeaways from this study are as follows:
- The most accessible/industrialized method of li-ion battery recycling is energy intensive and with relatively low amounts of mineral recovery
- The most accessible/industrialized method of li-ion battery recycling is only technically feasible & economical for select li-ion subchemistries
- Higher yield recycling methods are generally more expensive and/or produce high amounts of toxic waste
- More sustainable Li-ion battery processes are currently still being researched or only available at laboratory-scale
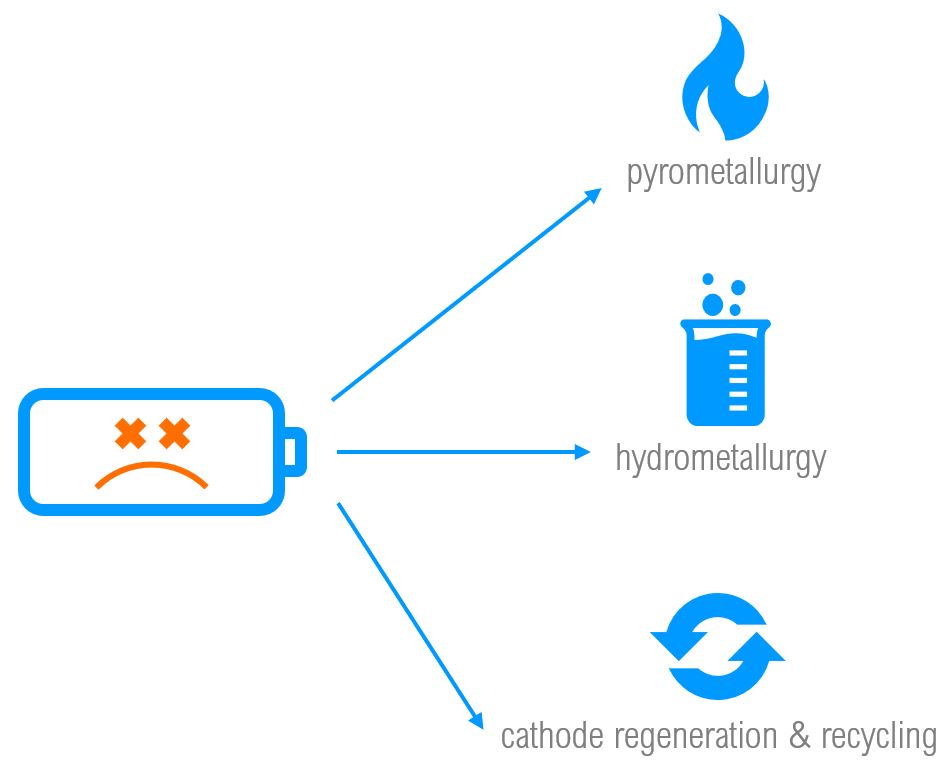
There are a few ways of recycling a Li-ion cells – we can use pyrometallurgical methods, hydrometallurgical methods, direct cathode regeneration/recycling, or combinations of all three. Simply speaking, pyrometallurgy involves extracting minerals using heat-based methods: calcinating, roasting, smelting, and refining. Hydrometallurgy involves extracting minerals using aqueous solutions through leaching, solution concentration/purification, and then electrolysis or precipitation. In many cases, Li-ion battery recycling plants use both pyrometallurgy and hydrometallurgy to obtain a greater, more diverse yield of recovered minerals from a Li-ion cell. The following chart summarizes key differences between pyrometallurgical and hydrometallurgical cell recycling methods:
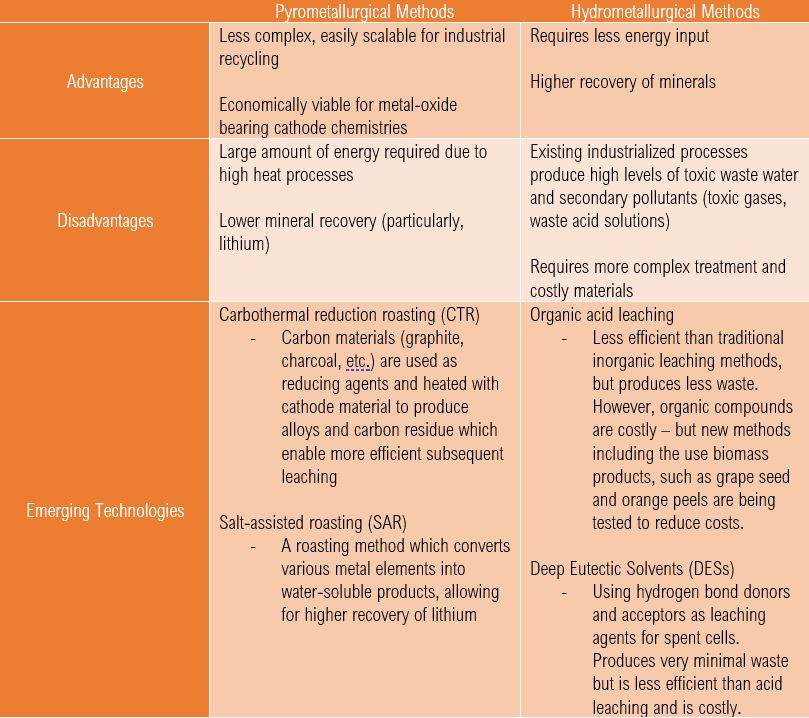
Hydrometallurgical methods generally have better metal recovery efficiencies than pyrometallurgical methods; however, traditional hydrometallurgical processes require more complex material separation and treatment, as well as much higher levels of water consumption and pollutants. Methods that improve these disadvantages (such as organic acid leaching and DESs leaching) require costly materials, and these methods only exist at laboratory-scale. Moreover, it is important to consider that different cell chemistries contain more/less valuable metals, which would affect the economic viability of recycling them altogether. For instance, lithium-iron phosphate cells (LFP) are not ideal candidates for pyrometallurgical recycling processes – which is the most accessible form of cell recycling today – as they are not comprised of high-cost metals such as cobalt and nickel. As a result, commercial recycling of LFP cells does not exist and the solutions we have today come at a high cost. However, as the commercial vehicle industry shifts towards using LFP cells due to their low-cost of fabrication and relatively safer and more stable operation, hydrometallurgical methods specific to the recycling of LFP cells may become more industrialized due to market pressure, therefore reducing costs and rendering it an economic process. Moreover, LFP cells are also contenders for cathode regeneration & recycling which is discussed in the following section.
Cathode Regeneration & Recycling
Regeneration of cathode materials is possible through combinations of pyrometallurgical & hydrometallurgical techniques. The conceptual benefit of regeneration is that rather than recovering bulk amounts of select materials through traditional recycling that are then sold back to manufacturers to be used in fabricating new products, regeneration aims to re-synthesize cell cathode materials all at once – keeping the circularity at the cell-level. This, however, eliminates the possibility of using recyclable cell materials in other applications – but reduces the number of steps in the li-ion recycling cycle. Disadvantages of current-day cathode regeneration techniques are that they require high level of input materials, intensive energy use, and generate high levels of waste and pollution (J.Mao et al. 2022). Additionally, only select cathode-types (NMC, LFP, LCO) have seen repeated success in laboratory settings. The feasibility of regeneration for other li-ion subchemistries has not been realized, and thus is far from industrialization. However, for LFP in particular, cathode regeneration could become a feasible method to recycle large amounts of spent cells in the future. Read more in the study by G. Ji et al. 2022: direct regeneration of degraded lithium-ion battery cathodes with a multifunctional organic lithium salt.
Cathode recycling, like cathode regeneration, also preserves circularity at the cell-level. Cathode recycling can be thought of as repairing a degraded cell, restoring its capacity to (or near) its original state, and is done via re-lithiation of the cathode – but can only be done to cells that have aged/degraded in a specific way (ie. degradation due to li-loss). There are several recycling methods – solid-state sintering, hydrothermal lithiation, molten salt lithiation, and electrochemical lithiation. Generally, solid-state sintering is most practical for industrialization; however, requires higher energy inputs than the other recycling methods.
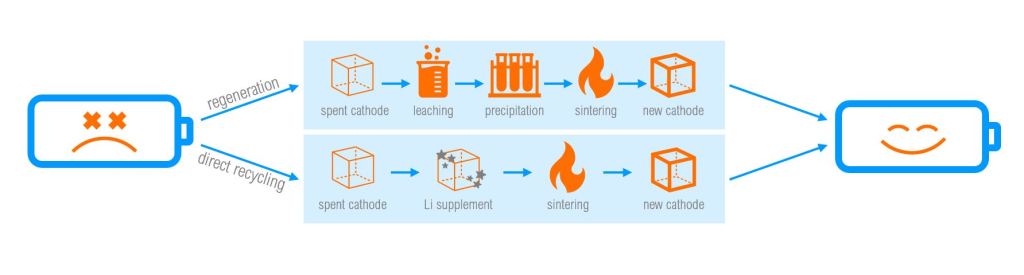
When comparing the two methods (regeneration vs. recycling), there are many pros and cons. Cathode regeneration is a more scalable process – although requires more complex syntheses and generates more waste and pollution; whereas, recycling through the re-lithiation of cell cathodes can handle materials with higher degradation level but requires more rigorous sorting and pre-processing of cell materials.
Is there anything else we can do to improve the life cycle of Lithium-Ion batteries?
Given the emergence of renewable-based energy such as wind and solar, there is an increasing demand in stationary energy storage solutions. Commercial vehicle batteries (as well as mining vehicle batteries) are often retired before their true end-of-life state, oftentimes still being capable of storing 60-70% of their initial capacities. Rather than being disposed of or sent for recycling, these batteries may still be utilized in lower-intensity applications, such as stationary energy-storage, if they are not damaged or faulty. According to this report by Mckinsey, “the second-life-battery supply for stationary application could exceed 200 gigawatt-hours per year by 2030. This volume will exceed the demand for lithium-ion utility scale storage for low- and high-cycle applications combined, which by 2030 will constitute a market with global value north of $30 billion”. The emerging market for second-life cells will help buy time for more economical recycling methods to emerge and industrialize.
An example of a second-life implementation by B2U Storage solutions can be found in Lancaster, California, where a 25 MWh of storage capacity made up of 1,300 retired Honda and Nissan EV batteries (some as much as 8 years old) is used to supplement a solar farm. For context, an average American household utilizes 29 kWh of energy a day – which means that in the event of the sun exploding, this storage implementation can supply energy to ~860 households for a period of 24 hours if fully “charged”. Ever neat! Of course, the actual impact of this solar farm depends on the size of the solar farm and available solar power, not just the capacity of the energy storage system.

So, what should we make of all this?
The answer nobody likes to hear – it depends. The energy required to produce a li-ion battery depends on the source of the minerals, the type of cell being fabricated, the design of the battery pack, the location of the manufacturer. The economic feasibility and outcome of recycling li-ion batteries depends on the type of cell and the type of processing available. The most environmentally friendly recycling methods are inaccessible (pilot scale only) and exceedingly expensive. The future popularity of Li-ion subchemistries depends on the type of battery-grade lithium being produced and their availability (LiOH vs Li2CO3). Moreover, all of this stops mattering when society begins to move on from lithium-based batteries altogether, which may happen sooner than we think.
What we do know is that industrialized recycling of all li-ion battery types has a long way to go; Particularly methods that are sustainable, energy non-intensive, and environmentally friendly.
Despite this post being primarily lithium-oriented, I anticipate that we will see the emergence of mineral recycling as a viable and necessary source of many electrification-supporting minerals (in addition to lithium) within my lifetime. However, further discussion of the critical minerals market requires a whole post in and of itself (and a lot more reading on my part).
Finally, perhaps pursuing a materials science degree with a focus on sustainable hydrometallurgical processing of batteries would have been a lucrative and stable career choice. Although, I suppose mining isn’t so bad either!
Further Questions to Explore
- This post is lithium-oriented, but how do these considerations change when considering other electrification-supporting materials (copper, nickel, iron, cobalt, manganese, etc.)
- Certain li-ion subchemistries are becoming more popular than others – how will this affect the mineral economical landscape (ie. will we move away from nickel-based li-ion cathodes?), and how will this influence industrial battery recycling?
- Recycling lithium-ion batteries may be energy intensive or produce waste – in what cases is raw lithium mining less destructive than recycling existing materials? How do expect this to change over time?
- What non-lithium-based batteries are emerging? What advancements in battery technology should we be looking forward to in the next few years? Will new battery technologies allow for better circularity as compared to lithium-ion batteries?
References
- EY, 2022. Critical minerals supply and demand challenges mining companies face.
- U.S. Geological Survey, 2022. Lithium Mineral Commodity Summary. https://www.usgs.gov/centers/national-minerals-information-center/lithium-statistics-and-information
- Internal Energy Agency, 2020. Global EV Outlook 2020. https://www.iea.org/reports/global-ev-outlook-2020
- C. Tabelin et al. 2021. Towards a low-carbon society: A review of lithium resource availability, challenges and innovations in mining, extraction and recycling, and future perspectives. Miner. Eng. 163, 106743.
- Businesswire, 2021. Compass Minerals Identifies Approximately 2.4 Million Metric Ton Sustainable Lithium Resource. https://www.businesswire.com/news/home/20210713006108/en/Compass-Minerals-Identifies-Approximately-2.4-Million-Metric-Ton-Sustainable-Lithium-Resource
- InsideEvs, 2022. Tesla’s 4680-Type Battery Cell Teardown: Specs Revealed. https://insideevs.com/news/598656/tesla-4680-battery-cell-specs/
- Battery University, 2019. BU-205: Types of Lithium-Ion. https://batteryuniversity.com/article/bu-205-types-of-lithium-ion
- J. Kelly et al, 2021. Energy, greenhouse gas, and water life cycle analysis of lithium carbonate and lithium hydroxide monohydrate from brine and ore resources and their use in lithium ion battery cathodes and lithium ion batteries. Resour Conserv Recy. 174, 105762.
- Innovation Origins, 2020. Producing gasoline and diesel emits more CO2 than we thought. https://innovationorigins.com/en/producing-gasoline-and-diesel-emits-more-co2-than-we-thought/
- Natural Resources Canada, 2023. 2023 Fuel Consumption Guide. https://natural-resources.canada.ca/energy-efficiency/transportation-alternative-fuels/fuel-consumption-guide/21002
- J. Mao et al, 2022. Toward practical lithium-ion battery recycling: adding value, tracking circularity and recycling oriented-design. Energy Environ. Sci. 15, 2732.
- G. Ji et al, 202. Direct regeneration of degraded lithium-ion battery cathodes with a multifunctional organic lithium salt. Nat Commun. 14, 584.
- Mckinsey, 2019. Second-life EV batteries: The newest value pool in energy storage. https://www.mckinsey.com/industries/automotive-and-assembly/our-insights/second-life-ev-batteries-the-newest-value-pool-in-energy-storage